 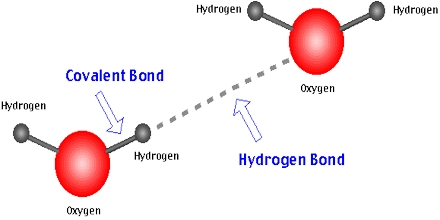
The two types of hydrogen bonding- intermolecular hydrogen bonding and intramolecular hydrogen bonding explain the type of molecule involved in a particular compound. Hydrogen bonding is known to explain different phenomena in the compounds. For example, the secondary forces operative in polyethylene are only van der waals forces, but their magnitude is so large that polyethylene is used to make bulletproof jackets. Long-chain polymers have stronger secondary forces than shorter ones. The mechanical properties of polymers, such as hardness, tensile strength, elasticity and melting point etc., depend upon the intermolecular forces between the polymer molecules. Alpha (α ) helical and beta (β) sheet structures of the protein are stable because of hydrogen bonding.Viscosity: Greater H-bonding leads to higher bonding between molecules, for example, glycerol.In DNA, H-bonding is present between bases.Ammonia (NH3) is soluble in water because of hydrogen bonding.Low concentrated alcohols are soluble in water due to hydrogen bonding.HCl and HBr exist in the form of gases due to the absence of hydrogen bonding.HF exists in the form of liquid because of hydrogen bonding.That is why it is present in the form of liquid, whereas van der waals force (weak) occurs between two molecules of H 2 S. In H 2 O, one water molecule is surrounded by four other water molecules by hydrogen bonding.H 2 O exist in the form of liquid, but H 2 S is present in the form of gas (though oxygen and sulphur belongs to the same group: 16).The hydrogen bonding should lead to forming either a five or six-membered ring, including the H-atom. /cdn.vox-cdn.com/imported_assets/1833217/Hydrogen_Bond_Quadruple_AngewChemIntEd_1998_v37_p75.jpg)
Simultaneously, the other group must also contain a highly electronegative atom and be further connected to an electronegative atom that is less reactive, for example, ortho-nitrophenol. The molecules should contain 02 groups so that one of the groups contains H-atoms connected to a more electronegative atom.The bonding of hydrogen atoms within a particular molecule is intramolecular hydrogen bonding, for example, salicylic acid.Ĭonditions for intramolecular hydrogen bonding: In H 2 O, one ( H 2 O ) molecule is surrounded by four other water molecules, and oxygen is joined by four bonds, two hydrogen bonds and two covalent bonds. When hydrogen bonding occurs among different types of molecules, either from the same or different compounds, it is called intermolecular bonding, for example, hydrogen fluoride, water, ammonia. Thus, only fluorine, oxygen, nitrogen atoms can form hydrogen bonds, as these atoms are small in size and have highly electronegative properties.The electronegative atom must be smaller in size.The molecules should contain a highly electronegative atom and be linked to H-atom.Conditions for Hydrogen Bonding in Polymers Hence, it is said to form a hydrogen bridge. A weak bond is formed among the two called Hydrogen Bond due to this electrostatic attraction.Īn H-atom links with two simultaneous electronegative atoms, one by hydrogen and another by covalent bonds. The positive end of a molecule attracts the negative end of another molecule. Hydrogen Bonding in Polymers (H-bonding) MeaningĪ hydrogen bond may be defined as the bond formed between a highly electronegative atom (such as fluorine, oxygen, nitrogen, sometimes chlorine) and a hydrogen atom. These intermolecular forces extend all along the polymer chain resulting in a significant combined effect. The polymer molecules are held together by weak secondary forces: van der waals forces, hydrogen bonding, and dipole-dipole interactions. It is tough but flexible and is used for making tires. Vulcanised rubber has cross-links of sulphur atoms. Cross-linking produces high melting polymers. This makes materials more rigid and less flexible. As a result, polymer molecules cannot slide over each other so easily. The primary forces may also join the polymer chains at different points such linkages are called cross-linking. Long or short polymer chains are formed depending upon the extent of covalent linkage in the polymer. In polymers, the atoms are joined by strong covalent bonds called primary forces. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
